How to check if your SAGA 32/64+ is a Medical device?
- All SAGA32/64+ purchased after 28/01/2021 are non-medical devices.
- All SAGA32/64+ SAGA32/64+ devices purchased within the EU region before 28/01/2021 are Medical Devices. Devices outside the EU region are non-medical devices.
If you do not know when your device was purchased. Check the following:
Step 1
Identify the Device Label (on the bottom of the Data recorder (or Docking Station).
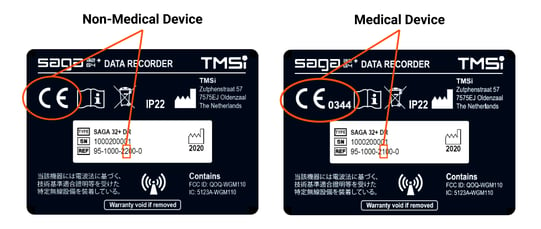
Step 2
Check the figure above. Is there a number adjacent to the CE marking: your device is a Medical Device. Is there NO number adjacent to the CE marking: your device is a Non-Medical Device.
Alternatively, check the REF code. Is the 8th number a '1': it is a medical device. Is the 8th number a '2': it is a non-medical device.
IMPORTANT!
Please note that the documentation and specifically the intended use for Medical and Non-Medical devices differ. Non-Medical devices are not to be used for medical purposes such as diagnosis, treatment or monitoring of vital physiological processes. No diagnostic or therapeutic conclusions may be drawn from the acquired signals. Download the documentation that is specific for your device to prevent misuse and misunderstanding.
In case of doubt: always contact support@tmsi.com for further information.
